In Part 5 of this series, we turn to the third major issue related to DNA contamination with residual bacterial plasmids and truncated mRNA from the manufacturing process. Are the vaccines more contaminated than our regulatory agencies realize? Should this raise concerns about migration to the gut or their expression by cells?
The questions posed throughout this series highlight the inherent safety issues associated with a lax regulatory framework for approval of the COVID-19 mRNA vaccines. In this article, we consider how lax regulation is related to DNA and RNA contamination issues.
Summary of Key Facts
- Concerns have been raised about DNA contamination in the mRNA COVID-19 vaccines. The specific concern is the presence of higher-than-expected residual DNA plasmids used in the original mRNA production. Independent investigations suggest that the Pfizer mRNA vaccine may have high levels of DNA contamination, potentially exceeding regulatory limits.
- There are theoretical risks associated with plasmid DNA expression and migration to the gut, which could affect human health and the microbiome. Concerns have also been raised about the quality control and manufacturing oversight of mRNA vaccines.
- The European Medicines Agency (EMA), Europe’s drug regulatory authority, noted the presence of truncated and modified RNA as impurities in the mRNA COVID-19 vaccines, raising the need for oversight.
- Related to the manufacturing process, a Danish study compared the rate of adverse events to the batch size (number of doses in a batch) and found a correlation.
The Advisory Committee on Immunization Practices met last week to recommend the updated COVID-19 vaccine. However, the manufacturers presented little data from testing in humans. Moderna was the only manufacturer to present safety and antibody response data from experience with 101 individuals. Pfizer presented antibody response data from 20 mice and is currently collecting data from 400 individuals in clinical testing. No data on manufacturing oversight was presented during the meeting.
As part of the safety evaluation of drug approval, the CMC (chemistry, manufacturing, and controls) process becomes critical in identifying and eliminating impurities. It sets strict standards and product specifications to maintain the purity of each batch. Compliance with these standards is essential for obtaining approval from global health authorities.
Imagine you’re a coffee drinker, and you decide to buy a bag of premium, freshly ground coffee beans from your favorite store. You expect that each bag contains pure, high-quality coffee grounds to brew that perfect cup of coffee. However, when you open the bag, you discover that it isn’t just coffee grounds; it also contains a mixture of sand and other foreign particles. This unexpected impurity completely ruins your experience.
Just as you rely on the purity of your coffee grounds for a great cup of coffee, the pharmaceutical industry, including vaccine production, has regulations in place to ensure good manufacturing practices. Patients and consumers expect that these guidelines mean that drug or vaccine formulations are free from unwanted substances, ensuring their safety and effectiveness.
Controlling impurities in traditional chemical products is a well-established practice, but for biological products such as mRNA-based vaccines, managing impurities presents unique challenges.
mRNA Products Contain ‘Gene Factories’
Double-stranded DNA (dsDNA) is used to make the mRNA contained in the COVID-19 vaccines. Tiny dsDNA plasmids are small engineered gene factories (Figure 1). These factories produce the mRNA strands contained in the LNPs. A plasmid appears like a tiny micro-bracelet with different segments representing different pieces of genes.
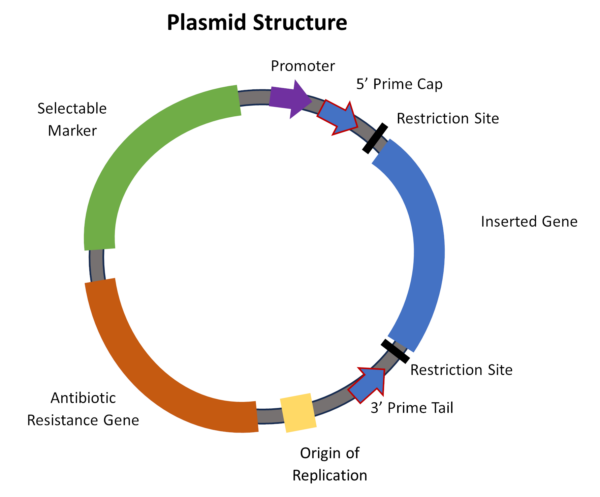
Regulatory agencies like EMA—Europe’s drug regulatory authority—set limits for the number of plasmids in the final lots distributed for injection. New questions have been raised about how much contamination there is, and whether the FDA is monitoring this. It is also unclear whether the plasmids can merge with human genes within the cell or travel to the gut.
The EMA standard for DNA contamination of vials is 0.33 percent (330 pg/mg), or roughly one DNA molecule for every 3,000 mRNA molecules. Although the Moderna mRNA-1273 vaccine meets this standard, the actual volume may be higher due to poor quality control. The DNA must be removed from the final product before distribution, but some residual amount is expected to remain. Unanswered questions include: How much DNA is in the vials? Is it over the limit? Is the FDA tracking this? And what are the implications, if any, with regard to persistence in the recipient?
There are at least two independent groups of investigators who have conducted lab tests and confirmed the mRNA vaccine of Pfizer has been contaminated by DNA.
One team of scientists, led by microbiologist Kevin McKernan, published a preprint paper showing that the Pfizer/BioNTech BNT162b2 vaccine has DNA “orders of magnitude higher than the EMA’s limit.” His paper has not yet been peer-reviewed. The batches examined, provided by an anonymous source, were unopened, expired vials that were not delivered on dry ice. If these data hold, the actual amount of plasmids was 18 to 70 times higher than the EMA limit. (Table 3, page 12.)
Obviously, future investigations should attempt to establish contamination levels using unexpired doses with an intact cold chain.
Professor Phillip Buckhaults, who holds a doctorate in biochemistry and molecular biology and is considered an expert in cancer genomics research at the University of South Carolina, performed an independent analysis for the presence of DNA in Pfizer batches.
Professor Buckhaults in testimony stated the following:
“The Pfizer vaccine is contaminated with plasmid DNA. It’s not just mRNA. It’s got bits of DNA in it. This DNA is the DNA vector that was used as the template for the in vitro transcription reaction when they made the mRNA. I know this is true because I sequenced it in my own lab.”
We will continue to follow this line of research.
Theoretical Risk of Plasmid DNA Contamination
While having some DNA in a sample is unavoidable and deemed acceptable, some have raised questions about the possibility for genomic integration of the DNA. Our cells use DNA in the nucleus to make protein, so if the plasmid DNA gets into the nucleus, there is a theoretical risk that it can get transcribed and make a protein.
About 5 to 10 percent of our human genome contains ancient retroviral DNA. However, this DNA is mutated to a point that is no longer harmful. Any further research on this topic will therefore need to establish not just the presence of DNA plasmid integration, but also its biological activity.
Professor Buckhaults further commented in his testimony:
“I am kind of alarmed about the possible consequences of this—both in terms of human health and biology—but you should be alarmed about the regulatory process that allowed it to get there.”
Concerns About DNA Migration to the Gut
Related to the DNA contamination is the concern about residual expression vectors, or plasmids, in the vials. To make a billion doses of mRNA vaccine, more than a kilogram of DNA is required. Plasmids help produce the DNA by splicing in the desired sequence into a bacterial plasmid (Figure 1).
Then workhorse bacteria, often E. coli, help spin out the DNA for production. These bacteria have an extra burden: They must replicate not only their own genome but also the plasmid DNA inserted within their genome. This takes slightly more time, so the bacteria without the additional DNA will eventually outcompete those with the DNA.
To solve this problem, scientists also splice in an antibiotic resistance gene. The entire pool of bacteria is then treated with an antibiotic to kill the faster-replicating bacteria without the conferred antibiotic resistance. This selective elimination allows the plasmid-carrying antibiotic-resistant bacteria to continue growing. In other words, this antibiotic resistance gene confers an advantage that drives selection pressure to favor the bacteria producing the desired DNA.
However, some scientists are concerned that exceeding EMA standards for DNA plasmid contamination could affect an already growing antibiotic resistance problem. This would be a potential concern only if plasmids containing the antibiotic resistance gene migrate to the gut, integrate with bacterial targets in the gut flora, and disrupt the microbiome of the gut accordingly. Diseases, including obesity, diabetes, cardiovascular disorders, cancer, hypertension, and irritable bowel syndrome have been loosely linked with disturbances of the gut microbiome.
Truncated mRNA Contamination
Nucleic acid contamination with truncated, or shortened, mRNA fragments is something that EMA has been following since February 2021. On page 35 of the EMA assessment report (pdf) on the BNT162b2 mRNA vaccine reviewed in Part 1, the EMA states, “Truncated and modified RNA are present as impurities.” The agency noted that the impurities were found at different levels during production. For instance, levels may be higher in smaller test batches than in larger commercial batches.
In fact, Danish scientists, Max Schmeling, Vibeke Manniche, and Peter Riis Hansen linked adverse events with vaccination records and found that smaller batches of the BNT162b2 mRNA vaccine may have a higher rate of adverse events (AEs). While this finding is intriguing, the authors call for more research to see if this is a consistent pattern. We reviewed the raw data provided by the authors and agree that a clustering of AEs seems to be found with batches having fewer than 100,000 doses.
[series_posts_list][/series_posts_list]
It has already been demonstrated in vitro in a laboratory experiment that mRNA can be reverse-transcribed to DNA within six hours. A remaining question is whether this can happen in a live organism. Thus far, there is no evidence that a reverse-transcribed DNA product can merge with a human cell’s genome. Claims about integration are solely speculative and based upon an evolutionary precedent for such a process.
The EMA asked for additional testing but allowed distribution to go forward. The scientists believed these fragments were unlikely to be intact mRNA fragments. An intact mRNA fragment needs to have a cap and a tail. The cap and tail are needed to tell the cell when to start and stop producing the spike protein.
Nevertheless, the EMA requested additional reports. The agency was concerned that an autoimmune reaction could be triggered if fragments’ potentially encoded proteins resemble human proteins. In other words, if the fragments “mimic” human proteins, antibodies could be developed against our own bodies.
“Any homology between translated proteins (other than the intended spike protein) and human proteins that may, due to molecular mimicry, potentially cause an autoimmune process should be evaluated. Due date: July 2021. Interim reports: March 2021, and on a monthly basis,” the EMA stated.
It is clear that the mass production of mRNA at an industrial scale carries potential risks. This issue has been raised recently by other researchers pointing out the role of manufacturing quality control. For instance, The Epoch Times has previously investigated the link between quality issues and clotting risk (Part 1, Part 2, and Part 4).
This issue of contamination by DNA and mRNA fragments should also be explored further to understand whether certain lots were affected more than others. We also need to know whether DNA contamination is linked to adverse events. The EMA should strictly follow its monitoring standards.
The issue of having any DNA contamination is biologically unavoidable given that the mRNA is transcribed from DNA vectors. The potential issue here is the unusually high level of DNA contamination involved in the mRNA vaccines.
However, pivoting these RNA therapeutics to a vaccine platform in the context of a lagging regulatory framework has left us with many unanswered questions. Public health officials, nevertheless, were adamant that this new product should be deployed in a one-size-fits-all manner ignoring differential COVID-19 risk profiles across a broad population. This, in turn, we believe, set the stage for policy overreach resulting in unethical and harmful mandates.
[note label=”Next” title=”Part 6 – FDA’s Failure to Safeguard the Public Resulted in Irreparable Erosion of Trust” hyperlink=”https://m.theepochtimes.com/health/fdas-failure-to-safeguard-the-public-resulted-in-an-irreparable-erosion-of-trust-5219480″ description=”In Part 6, we will discuss whether the FDA should have eased mRNA vaccine regulations during the pandemic and the safety consequences of regulatory delays.”][/note]
◊ References
Alana F Ogata, Chi-An Cheng, Michaël Desjardins, Yasmeen Senussi, Amy C Sherman, Megan Powell, Lewis Novack, Salena Von, Xiaofang Li, Lindsey R Baden, David R Walt, Circulating Severe Acute Respiratory Syndrome Coronavirus 2 (SARS-CoV-2) Vaccine Antigen Detected in the Plasma of mRNA-1273 Vaccine Recipients, Clinical Infectious Diseases, Volume 74, Issue 4, 15 February 2022, Pages 715–718, https://doi.org/10.1093/cid/ciab465
Aldén M, Olofsson Falla F, Yang D, Barghouth M, Luan C, Rasmussen M, De Marinis Y. Intracellular Reverse Transcription of Pfizer BioNTech COVID-19 mRNA Vaccine BNT162b2 In Vitro in Human Liver Cell Line. Curr Issues Mol Biol. 2022 Feb 25;44(3):1115-1126. doi: 10.3390/cimb44030073. PMID: 35723296; PMCID: PMC8946961. https://pubmed.ncbi.nlm.nih.gov/35723296/
Allergic Reactions Including Anaphylaxis After Receipt of the First Dose of Pfizer-BioNTech COVID-19 Vaccine — United States, December 14–23, 2020. MMWR Morb Mortal Wkly Rep 2021;70:46–51. DOI: http://dx.doi.org/10.15585/mmwr.mm7002e1
Anderson EJ, Rouphael NG, Widge AT, et al. Safety and Immunogenicity of SARS-CoV-2 mRNA-1273 Vaccine in Older Adults N Engl J Med 2020; 383:2427-2438 https://www.nejm.org/doi/full/10.1056/NEJMoa2028436
Anderson S. CBER Plans for Monitoring COVID-19 Vaccine Safety and Effectiveness. https://stacks.cdc.gov/view/cdc/97349 October 20, 2020. Accessed 3/20/23.
Angeli F, Spanevello A, Reboldi G, Visca D, Verdecchia P. SARS-CoV-2 vaccines: Lights and Shadows. Eur J Intern Med. 2021 Jun;88:1-8. doi: 10.1016/j.ejim.2021.04.019. Epub 2021 Apr 30. PMID: 33966930; PMCID: PMC8084611. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC8084611/#bib0043
Baker, A. T., Boyd, R. J., Sarkar, D., Teijeira-Crespo, A., Chan, C. K., Bates, E., Waraich, K., Vant, J., Wilson, E., Truong, C. D., Lipka-Lloyd, M., Fromme, P., Vermaas, J., Williams, D., Machiesky, L., Heurich, M., Nagalo, B. M., Coughlan, L., Umlauf, S., Chiu, P. L., … Borad, M. J. (2021). ChAdOx1 interacts with CAR and PF4 with implications for thrombosis with thrombocytopenia syndrome. Science Advances. 7(49), eabl8213. https://doi.org/10.1126/sciadv.abl8213
Baumeier C, Aleshcheva G, Harms D, Gross U, Hamm C, Assmus B, Westenfeld R, Kelm M, Rammos S, Wenzel P, Münzel T, Elsässer A, Gailani M, Perings C, Bourakkadi A, Flesch M, Kempf T, Bauersachs J, Escher F, Schultheiss H-P. Intramyocardial Inflammation after COVID-19 Vaccination: An Endomyocardial Biopsy-Proven Case Series. International Journal of Molecular Sciences. 2022; 23(13):6940. https://doi.org/10.3390/ijms23136940
Bloom, K., van den Berg, F. & Arbuthnot, P. Self-amplifying RNA vaccines for infectious diseases. Gene Ther 28, 117–129 (2021). https://doi.org/10.1038/s41434-020-00204-y.
Chauhan, H., Mohapatra, S., Munt, D.J. et al. Physical-Chemical Characterization and Formulation Considerations for Solid Lipid Nanoparticles. AAPS PharmSciTech 17, 640–651 (2016). https://doi.org/10.1208/s12249-015-0394-x
Chui CSL, Fan M, Wan EYF, et al. Thromboembolic events and hemorrhagic stroke after mRNA (BNT162b2) and inactivated (CoronaVac) covid-19 vaccination: A self-controlled case series study. Lancet. 2022;(50). https://www.thelancet.com/journals/eclinm/article/PIIS2589-5370(22)00234-6/fulltext
Dag Berild J, Bergstad Larsen V, Myrup Thiesson E, et al. Analysis of Thromboembolic and Thrombocytopenic Events After the AZD1222, BNT162b2, and MRNA-1273 COVID-19 Vaccines in 3 Nordic Countries. JAMA Netw Open. 2022;5(6):e2217375. doi:10.1001/jamanetworkopen.2022.17375 https://jamanetwork.com/journals/jamanetworkopen/fullarticle/2793348
daSilva RL. Viral-associated thrombotic microangiopathies. Hematology/Oncology and Stem Cell Therapy. 2011:4(2):51-59. https://www.sciencedirect.com/science/article/pii/S165838761150038X
De A, Ko YT. Why mRNA-ionizable LNPs formulations are so short-lived: causes and way-out. Expert Opin Drug Deliv. 2023 Feb;20(2):175-187. doi: 10.1080/17425247.2023.2162876. Epub 2023 Jan 1. PMID: 36588456. https://pubmed.ncbi.nlm.nih.gov/36588456/
Ehaideb, S.N., Abdullah, M.L., Abuyassin, B. et al. Evidence of a wide gap between COVID-19 in humans and animal models: a systematic review. Crit Care 24, 594 (2020). https://doi.org/10.1186/s13054-020-03304-8
European Medicines Agency (pdf)
Faizullin D, Valiullina Y, Salnikov V, Zuev Y. Direct interaction of fibrinogen with lipid microparticles modulates clotting kinetics and clot structure. Nanomedicine. 2020 Jan;23:102098. doi: 10.1016/j.nano.2019.102098. Epub 2019 Oct 23. PMID: 31655206. https://pubmed.ncbi.nlm.nih.gov/31655206/
FDA. Considerations for Human Radiolabeled Mass Balance Studies – Guidance for Industry. https://www.fda.gov/media/158178/download May, 2022.
FDA. Development and Licensure of Vaccines to Prevent COVID-19. https://www.fda.gov/media/139638/download
FDA-CBER-2021-5683-0013962 approved on: 09-Nov-2020. A Tissue Distribution Study of a [3H]-Labeled Lipid Nanoparticle-mRNA Formulation Containing ALC-0315 and ALC-0159 Following Intramuscular Administration in Wistar Han Rats. FINAL REPORT Test Facility Study No. 185350 Sponsor Reference No. ALC-NC-0552 (pdf)
Fertig TE, Chitoiu L, Marta DS, Ionescu VS, Cismasiu VB, Radu E, Angheluta G, Dobre M, Serbanescu A, Hinescu ME, Gherghiceanu M. Vaccine mRNA Can Be Detected in Blood at 15 Days Post-Vaccination. Biomedicines. 2022 Jun 28;10(7):1538. doi: 10.3390/biomedicines10071538. PMID: 35884842; PMCID: PMC9313234. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC9313234/
Grobbelaar LM et al. SARS-CoV-2 spike protein S1 induces fibrin(ogen) resistant to fibrinolysis: implications for microclot formation in COVID-19 Biosci Rep (2021) 41 (8): BSR20210611. https://doi.org/10.1042/BSR20210611
Hou, X., Zaks, T., Langer, R. et al. Lipid nanoparticles for mRNA delivery. Nat Rev Mater 6, 1078–1094 (2021). https://doi.org/10.1038/s41578-021-00358-0
Iaboni A, Jindal N, Betschel SD, Song C. Second-dose mRNA COVID-19 vaccine safety in patients with immediate reactions after the first dose: A case series. Journal of Allergy and Clinical Immunology: Global. 2022;1(3): 172-174. https://www.sciencedirect.com/science/article/pii/S2772829322000200
Let’s talk about lipid nanoparticles. Nat Rev Mater 6, 99 (2021). https://www.nature.com/articles/s41578-021-00281-4
Li, JX., Wang, YH., Bair, H. et al. Risk assessment of retinal vascular occlusion after COVID-19 vaccination. npj Vaccines 8, 64 (2023). https://doi.org/10.1038/s41541-023-00661-7
Michieletto, D., Lusic, M., Marenduzzo, D. et al. Physical principles of retroviral integration in the human genome. Nat Commun 10, 575 (2019). https://doi.org/10.1038/s41467-019-08333-8
Moghimi, S. M., & Simberg, D. (2022). Pro-inflammatory concerns with lipid nanoparticles. Molecular therapy : The Journal of the American Society of Gene Therapy, 30(6), 2109–2110. https://doi.org/10.1016/j.ymthe.2022.04.011
Naturally Inspired Podcast. Jessica Rose PhD – VAERS, Data And Truth https://www.audible.com/pd/Jessica-Rose-PhD-VAERS-Data-And-Truth-Podcast/B09YMLJGBN?clientContext=132-5166709-6339436&loginAttempt=true&noChallengeShown=true
Ohlson J. Plasmid manufacture is the bottleneck of the genetic medicine revolution. Drug Discov Today. 2020 Oct 16;25(11):1891–3. doi: 10.1016/j.drudis.2020.09.040. Epub ahead of print. PMID: 33075470; PMCID: PMC7564888. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC7564888/
Perico L, Marina Morigi M, Galbusera M, et al. SARS-CoV-2 Spike Protein 1 Activates Microvascular Endothelial Cells and Complement System Leading to Platelet Aggregation. Front. Immunol. 2022 https://www.frontiersin.org/articles/10.3389/fimmu.2022.827146/full
Qin, S., Tang, X., Chen, Y. et al. mRNA-based therapeutics: powerful and versatile tools to combat diseases. Sig Transduct Target Ther 7, 166 (2022). https://doi.org/10.1038/s41392-022-01007-w
Rafati A, Pasebani Y, Jameie M, et al. Association of SARS-CoV-2 Vaccination or Infection With Bell Palsy: A Systematic Review and Meta-analysis. JAMA Otolaryngol Head Neck Surg. Published online April 27, 2023. doi:10.1001/jamaoto.2023.0160 https://jamanetwork.com/journals/jamaotolaryngology/fullarticle/2804297
Rhea EM, Logsdon AF, Hansen KM, Williams LM, Reed MJ, Baumann KK, Holden SJ, Raber J, Banks WA, Erickson MA. The S1 protein of SARS-CoV-2 crosses the blood-brain barrier in mice. Nat Neurosci. 2021 Mar;24(3):368-378. doi: 10.1038/s41593-020-00771-8. Epub 2020 Dec 16. PMID: 33328624; PMCID: PMC8793077. https://pubmed.ncbi.nlm.nih.gov/33328624/
Röltgen K, Nielsen SCA, Silva O. Immune imprinting, breadth of variant recognition, and germinal center response in human SARS-CoV-2 infection and vaccination. Cell 2022;185(6):1025-1040. https://www.cell.com/cell/fulltext/S0092-8674(22)00076-9
Rosenblum HG, Gee J, Liu R, et al. Safety of mRNA vaccines administered during the initial 6 months of the US COVID-19 vaccination program: an observational study of reports to the Vaccine Adverse Event Reporting System and v-safe. Lancet Infectious Diseases. 2022;22(6):802-812. https://doi.org/10.1016/S1473-3099(22)00054-8
Schmeling, M, Manniche, V, Hansen, PR. Batch-dependent safety of the BNT162b2 mRNA COVID-19 vaccine. Eur J Clin Invest. 2023; 00:e13998. doi:10.1111/eci.13998
Srinivasan M, Thangaraj SR, Arzoun H. Gene Therapy – Can it Cure Type 1 Diabetes? Cureus. 2021 Dec 19;13(12):e20516. doi: 10.7759/cureus.20516. PMID: 35004071; PMCID: PMC8723777. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC8723777/
Trougakos IP, Terpos E, Alexopoulos H, et al. Adverse effects of COVID-19 mRNA vaccines: the spike hypothesis. Cell 2022;28(7): P542-554. https://www.cell.com/trends/molecular-medicine/fulltext/S1471-4914(22)00103-4
Vervaeke P, Borgos SE, Sanders NN, Combes F. Regulatory guidelines and preclinical tools to study the biodistribution of RNA therapeutics. Adv Drug Deliv Rev. 2022 May;184:114236. doi: 10.1016/j.addr.2022.114236. Epub 2022 Mar 26. PMID: 35351470; PMCID: PMC8957368. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC8957368/
Wong DWL, Klinkhammer BM, Djudjaj S, Villwock S, Timm MC, Buhl EM, Wucherpfennig S, Cacchi C, Braunschweig T, Knüchel-Clarke R, Jonigk D, Werlein C, Bülow RD, Dahl E, von Stillfried S, Boor P. Multisystemic Cellular Tropism of SARS-CoV-2 in Autopsies of COVID-19 Patients. Cells. 2021 Jul 27;10(8):1900. doi: 10.3390/cells10081900. PMID: 34440669; PMCID: PMC8394956.
Yonker LM, Swank Z, Bartsch YC, et al. Circulating Spike Protein Detected in Post COVID-19 mRNA Vaccine Myocarditis. Circulation. 2023:147(11). https://www.ahajournals.org/doi/10.1161/CIRCULATIONAHA.122.061025
Views expressed in this article are the opinions of the author and do not necessarily reflect the views of The Epoch Times. Epoch Health welcomes professional discussion and friendly debate. To submit an opinion piece, please follow these guidelines and submit through our form here.